
Keywords: Mercury resistance (mer) operon, sulfhydrylase, oxidateve stress response, Thermus thermophilus
Mercury resistance and the oxidtive stress response, which prevents intracellular oxygen damage, share dependence on the availability of cysteine. We are investigating how the supply of thiolated amino acids affects resistance to mercury and to reactive oxygen species (ROS) with the goal of learning how metals and oxidative stresses interact intracellularly.

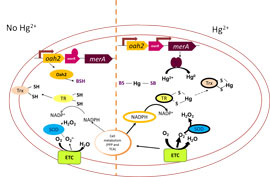
When mercury (Hg) is not present in the cell environment, the Hg resistance (mer) operon is partially repressed expressing only MerR, the repressor of the operon, and Oah2, an enzyme involved in redox buffer biosynthesis. MerA, the enzyme that reduces and detoxifies Hg, is not made due to a tight repression of the merA promoter by MerR.
When Hg enters the cells, it is rapidly sequestered by the small molecule redox buffers such as bacilithiol (BSH). This challenges the thiol homeostasis, destabilizing the intracellular redox state leading to a higher requirement for electrons and thus increased respiration rates and oxidative stress. However, Hg also increases expression (thick lines) of the mer operon. This induction leads to production of more Oah2 and thus more BSH and of MerA and the detoxification of Hg by reduction to the volatile elemental form.


